Steps of Oxidative Phosphorylation
Introduction
Oxidative phosphorylation is a process by which the energy-rich molecules ATP and NADH are produced in cells. It occurs within the mitochondria and is used to generate ATP in a form that can be used to drive cellular bioenergetics.
This process is the final step of cellular respiration and occurs in the inner membrane of the mitochondrion. It is a process that uses various electron acceptors such as oxygen and allows for the transfer of electrons into a transport chain.
The Mitochondrion Structure


The mitochondrion is a double membrane organelle. The outer membrane contains protein complexes, and the inner membrane has enzymes involved in oxidative phosphorylation. Several channels allow for the transport of molecules from one side to another. Mitochondria have three main parts, namely the matrix, intermembrane space, and the inner membrane.
The matrix is located between the outer membrane and the inner membrane; it contains ribosomes involved in protein synthesis. The intermembrane space is found between the inner and outer membranes where high-energy electrons from NADH and FADH2 pass through to create ATP. The inner membrane is smooth, and porin protein complexes are found throughout it.
The mitochondrial matrix contains enzymes responsible for creating some components of the respiratory chain, ATP synthase, and other ATP-producing proteins. The inner membrane and its channel system are involved in the transport of various molecules between the two membranes
The Role of Oxidative Phosphorylation in Cellular Respiration


Oxidative phosphorylation is an essential mechanism in cellular respiration. Cellular respiration refers to various chemical reactions within the cell. These reactions use oxygen as part of the electron transport chain.
This process converts adenosine diphosphate (ADP) into adenosine triphosphate (ATP), the primary energy source for all living organisms. Oxidative phosphorylation produces ATP through the chemical reactions of the mitochondrion.
How cellular respiration happens
Glycolysis. It takes place in the cytoplasm and involves the breakdown of glucose to pyruvate and generates a net gain of 2 ATP molecules per glucose molecule. The enzymes involved are phosphofructokinase, enolase, and pyruvate kinase.


Glycolysis takes place in the cytoplasm. ATP is used to phosphorylate glucose. It causes one glucose molecule to break down into two molecules of pyruvate and produces two ATP molecules per glucose molecule. The net gain in glycolysis is 2 ATP and 2 NADH…
Macromolecules are not usually produced from the breakdown of glucose. However, if there is not enough oxygen in the cell during glycolysis, pyruvate can be converted into lactate. Lactate molecules provide energy for the cell instead of ATP. The end product of glycolysis is pyruvate.
Krebs cycle. This step occurs in the mitochondrion, and through a process called oxidative phosphorylation, it generates more ATP than glycolysis. It also produces 32 molecules of NADH along with 20 molecules of FADH2.


In the Krebs cycle, glucose is broken down to produce 2 carbon fragments. These are then oxidized and combined with coenzymes.
Electron transport chain. It generates a reduced form of NADH which contains high-energy electrons that can be used in the phosphorylation stage to create ATP.
Phosphorylation stage. ATP is generated from ADP and pyrophosphate in the presence of oxygen.
The Significance of the Citric Cycle/Kreb’s Cycle in Phosphorylation Oxidation
During the citric acid cycle, NADH and FADH2 are oxidized through a process called oxidative phosphorylation. This results in the creation of ATP and water. The citric acid cycle takes place in the mitochondria.
Planck’s equation shows that the amount of energy given off is proportional to the frequency with which photons are emitted and their wavelength. Hence, when a molecule absorbs photon energy, it changes its original vibrational quantum state. This leads to a shift in its energy level. A different energy level means a change of state from the original ground state to an excited state.


The maximum wavelength of light is 545 nm, and this corresponds with the color yellow. When sunlight hits plants, chlorophyll molecules absorb yellow photons and are excited to a high-energy quantum state because of their high light absorption. Excited chlorophyll molecules start a chain of chemical reactions that build sugars from carbon dioxide and water.
The Main Stages of Oxidative Phosphorylation
Oxidative phosphorylation occurs in two main steps; firstly, electron carriers bring the high-energy electrons from the mitochondrial matrix across the membrane to a site known as complex IV. This is the NADH and FADH2 oxidation.
In the second step, some electrons are transferred down a set of electron carriers to coenzyme Q10 and then onto NADH. This is the phosphorylation stage.
The electrons are then accepted by a different carrier known as cytochrome c, passing them into the inner mitochondrial membrane. Here they are accepted by complex III. The electrons then pass through a chain of carriers to oxygen and form the final electron acceptor.
Additionally, the second transfer of electrons occurs within complex III, where some high-energy electrons are transferred to NADH. These electrons are then shuttled across the membrane to complex I, passed on to coenzyme Q10. The final product of this process is the generation of ATP from ADP and inorganic phosphate.
1. The NADH and FADH2 Oxidation Stage


It is the step where electron transfer happens in the inner membrane of the mitochondria and is where most ATP is made. This stage uses NADH and FADH2, which are transported across the inner membrane by coenzyme Q10. Here they are combined with the high-energy electrons and then passed to cytochrome C.
The oxidation stage happens in four main steps:
- In the first step of oxidation, the citric acid cycle releases three NADH molecules and one FADH2 molecule into the inner membrane. They are then oxidized by three high-energy electrons, where one is taken from each NADH and FADHT2, meaning that two electrons have the potential to be used in later steps.
- The second step is proton pumping via protein complex I or NADH dehydrogenase. This process allows the pumping of protons from the inner to the outer membrane through a chain of carriers and creates a proton gradient.


The electrons received from the first step pass through a series of carriers and move from enzyme to enzyme along their transport chain. Here they are passed down via electron carriers. This results in a proton gradient and the potential to create ATP with the addition of ADP and phosphate.
Electron transfer is repeated in the third step. It is where electrons pass from complex I to complex III via coenzyme Q10.
- In the third step, electrons are transferred to complex III via coenzyme Q10. Here, a second proton gradient is created. The energy released through this process is used to create ATP.
- In the final step of the oxidation stage, coenzyme Q10 attaches to cytochrome C, and the electrons are shuttled across to complex IV and onto oxygen. This process generates three ATPs from each pair of high-energy electrons.
2. The Phosphorylation Stage
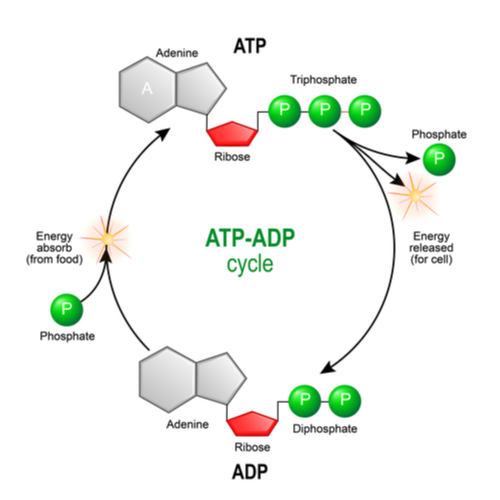

Phosphorylation is the final stage of the mitochondrial pathway and this process is where ATP is generated. This step involves the oxidation of NADH by cytochrome C and the addition of one electron to create a molecule known as a hydrogen ion or hydroxyl radical. The hydrogen ions are pumped into the inner membrane by complex IV, also known as cytochrome C oxidase.
To increase efficiency in the production of ATP, complex V or F1Fo-ATP synthase is used. Complex V adds a molecule of inorganic phosphate to ADP, allowing for the release of stored energy within ATP. This is the stage where the majority of the ATP is made.
Not all these high-energy electrons are used in ATP synthesis. Some of the energy is lost during this process. Some energy is also lost as heat, and some of it is used to transport the high-energy electrons. This means that less than 36 ATP molecules can be made from one electron. The rest is only made from the energy in the hydroxyl radical.
Many stages are included to produce the most amount of ATP from NADH and FADH2. The oxidative phosphorylation occurs during aerobic respiration, and that a total of 36 ATP molecules are made. This is because each molecule of a high-energy electron can be used to produce 3 ATPs.


The high-energy molecules are passed from one carrier to another via a complex transport system. The NADH and FADH2 start as hydrogen ions which then become NAD+ and FAD. The three high-energy electrons then oxidize these to produce water. This creates the hydroxyl radical in the phosphorylation stage. Finally, the three ATPs are made.
The Use of Oxygen in Oxidative Phosphorylation
The first thing that is important to know about oxygen is that oxidative phosphorylation must occur. This stage of respiration requires O2, as this is where the oxidation process takes place. Oxygen also helps determine which metabolic pathway will be used.
One reason why O2 is needed is that it is the final electron acceptor in the respiratory chain. Once this has occurred, ATP can be synthesized to release energy for use in the cell.
Using O2 means that one molecule of O2 is used upper two molecules of NADH or FADH2, resulting in a much higher yield than if it was not used. It is also easier for the cells to transport oxygen than for them to transport NADH or FADH2. The latter molecules are pretty large.
Some things need to take place for oxidative phosphorylation to occur, and all these require the presence of O2: The electrons from NADH have to pass through several steps via coenzyme Q10 and end up in complex III. Complex IV or cytochrome C oxidase has to transfer its electrons to O2 then release H+ ions via an electron transport chain. The final stage is where ATP is synthesized.
Synthase mechanisms, on the other hand, do not use oxygen. It involves the synthesis of ATP and is done by substrate-level phosphorylation. This process involves the transfer of phosphate from high-energy molecules to ADP, resulting in ATP being synthesized and released as a product.


The ATP Molecules
ATP molecules are made in the last stage of oxidative phosphorylation, and they are made during aerobic respiration.
Aerobic respiration takes place in the mitochondria, and it is where the majority of ATP is made. There are a set number of stages that convert NADH and FADH2 into ATP molecules.
Transporting the high-energy electrons to oxygen is where most of the ATP molecules are produced, while the rest is made due to this process. The byproducts are waste such as carbon dioxide and water.
The energy stored in the electrons is converted into ATP molecules when inorganic phosphate is added to ADP to create an ester bond. This addition results in the storage of the energy within ATP. The ATP molecules are used for chemiosmosis, muscle contraction, and transport of substances across cell membranes.
How does the ATP Yield Affect Oxidation Phosphorylation?
The yield of ATP affects the rate of oxidative phosphorylation, which is how fast the entire process occurs. The higher the energy released, the faster the process occurs and vice versa. This is because the higher the number of ATP molecules, the more energy is generated within the mitochondria.
The amount of energy produced varies depending on the energy of the electrons from NADH and FADH2. As a result, mitochondria convert these two molecules into different amounts of ATP. The more energy that is released, the higher the number of ATP molecules produced from each NADH and FADH2.
For example, if the yield of ATP from each molecule NADH is 2, then there will be 2 ATP molecules produced. However, if the energy is increased to 4, the number of ATP molecules will double to 4. Some electrons release a lot more energy, producing large amounts of ATP from only small amounts of high-energy electrons.
How does ATP get recycled?
ATP is a critical molecule in biological systems because it provides energy for the majority of biochemical reactions. It is made during periods of high-energy demand when oxygen levels are plentiful. The molecule is used at times of low-energy need when oxygen levels are low. It gets recycled by two different processes:
1) Phosphorylation, and
2) Degradation.


Phosphorylation involves the addition of a phosphate group to ADP and causes it to become ATP again. This process occurs in the mitochondria during the process of oxidative phosphorylation.
To degrade ATP, enzymes break it down into adenosine monophosphate (AMP) and inorganic phosphate.
The Electron Transport Chain


The electron transport chain is a series of carriers that carry the electrons from NADH and FADH2. There are five separate components to this system, and they all connect to form a complex network.
The electron transport chain is located in the inner membrane of the mitochondria, and this is where oxidative phosphorylation occurs.
The five components of the electron transport chain are:
1) NADH dehydrogenase,
2) ubiquinone,
3) cytochrome C,
4) cytochrome C reductase
5) cytochrome C oxidase.
NADH Dehydrogenase


NADH dehydrogenase is a protein containing 2 iron atoms in its active site capable of accepting electrons. It includes flavin adenine dinucleotide (FAD) in its structure. A FAD is a molecule that carries 2 electrons.
This enzyme transfers the high-energy electrons from NADH to FAD, and it produces a hydrogen ion as a byproduct. This is the first step in oxidative phosphorylation, and it allows for the production of ATP.
Ubiquinone


Ubiquinone transfers the electrons from the NADH dehydrogenase to the next molecule. It is also known as coenzyme Q10, as it contains 10 carbon atoms.
The electrons are transferred from NADH to ubiquinone, and this then transfers them to the third molecule. This step releases a hydrogen ion as a byproduct.
Cytochrome C


It is bound to the inner membrane of the mitochondria and transfers electrons from ubiquinone to cytochrome C reductase. The electrons then enter the final component of the electron transport chain. Cytochrome C acts as a “pump,” and it allows electrons to pass through the membrane.
In eukaryotes, cytochrome C is found inside the mitochondria, and it acts as an electron carrier. It has no cell of its own and remains soluble within the lipid bilayer of the mitochondrion.
Cytochrome C Reductase


The cytochrome C reductase transfers the electrons from cytochrome C to cytochrome oxidase. It also contains a FAD which is the same as NADH dehydrogenase.
The electrons are transferred to the next stage of oxidative phosphorylation, and this results in a hydrogen ion that is a byproduct. As the electrons move down this chain, they lose energy and become more stable.
Cytochrome C Oxidase


The final and last part of the electron transport chain is cytochrome C oxidase. It is a protein that contains a heme group, and this then transfers the electrons to oxygen, creating water in the process. All the NADH and FADH2 have been turned into water to create ATP molecules, the byproducts of the process.
The oxidation of NADH and FADH2 produces a total of 3 ATP molecules and 2 H+ ions. The process of oxidative phosphorylation is an essential part of respiration, and it provides more ATP molecules than glycolysis.
The electron transport chain creates a proton gradient used as the energy source for glycolysis and other reactions.
What is Chemioisis in Oxidative Phosphorylation?


Chemioisis is the creation of ATP by substrate-level phosphorylation. It means that energy can be produced without oxygen, and it occurs due to a proton gradient.
The proton pumps in the electron transport chain create the proton gradient, and this is used by ATP synthase to make ATP. The protons are transported through the inner membrane of the mitochondria, and this produces ATP.
The proton pump in oxidative phosphorylation is similar to the F1F0 complex in glycolysis as both complexes have a proton gradient.
As cytochrome C oxidase transfers the electrons to oxygen, it regenerates ubiquinone. This regeneration allows for the start of another round of chemioisis.
In the inner mitochondrial membrane, the ATP synthase is located, and this is where ATP is created. The enzyme uses the proton gradient created by the electron transport chain to produce ATP molecules.
If there was a proton gradient in glycolysis, the F1F0 complex would not be necessary as ATP synthase is already present.
Electrochemical gradient and chemioisis


The electrochemical gradient and chemioisis are related in the sense that it is like a fuel for ATP. This means that energy can be produced from the oxidative phosphorylation of metabolic processes without oxygen being required.
In terms of oxidative phosphorylation, protons are pumped into the inner membrane by complex IV, and the enzyme ATP synthase produces ATP. This creates energy.
The chemiosmotic hypothesis of Peter Mitchell was his theory that the proton gradient created in oxidative phosphorylation acted as a primary source of energy for the synthesis of ATP in mitochondria, chloroplasts, and bacteria. Then when oxygen is not required to create energy, this is called chemioisis.
In Respiratory Acidosis and Alkalosis, the pH of the blood can become too acidic. Increased carbon dioxide levels in the blood, such as in respiratory acidosis, will allow for more bicarbonate ions to be produced by the red blood cells. These are transported into the blood and the lungs. This is why increasing bicarbonate levels can treat respiratory acidosis in the blood with sodium bicarbonate. In severe cases, dialysis is applicable.
The Proton Pumping Complex
This is the part of the electron transport chain responsible for pumping protons, and it is the first stage of oxidative phosphorylation. This step is only essential in creating a proton gradient, and this proton gradient makes ATP synthesis possible.
This step involves the oxidation of NADH by cytochrome C (complex I) and the addition of electrons to create a hydrogen ion or hydroxyl radical. These hydrogen ions are pumped into the inner membrane by complex IV, also known as cytochrome C oxidase.
The proton pumping complex consists of five protein subunits, A-E, located in the mitochondria’s inner membrane. It also contains heme groups from cytochrome C, which is a part of the electron transport chain.
These protein subunits are located in the inner membrane, and they transfer the electrons from NADH to complex III, also known as ubiquinone. The electrons are then transferred to complex IV or cytochrome C oxidase, which pumps the protons across the inner membrane.
You may be interested in chromatins
Summary
The process of oxidative phosphorylation is a complex one, but it can be broken down into the following steps. First, ATP synthase takes in ADP and releases energy. Next, NADH dehydrogenase removes electrons from NAD+ to produce more FADH2, entering the electron transport chain.
Finally, cytochrome C oxidase (COX) accepts two hydrogen ions and oxygen atoms while releasing H 2 O molecules that we breathe out as waste products. If you want to learn more about this topic or how these mechanisms work together, read more of our cellular respiration blogs.


I‘m a freelance content and SEO writer with a passion for finding the perfect combination of words to capture attention and express a message. I create catchy, SEO-friendly content for websites, blogs, articles, and social media. My experience spans many industries, including health and wellness, technology, education, business, and lifestyle. My clients appreciate my ability to craft compelling stories that engage their target audience, but also help to improve their website’s search engine rankings. I’m also an avid learner and stay up to date on the latest SEO trends. I enjoy exploring new places and reading up on the latest marketing and SEO strategies in my free time.







